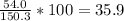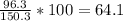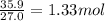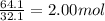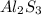Chemistry, 20.10.2019 09:50 strange5eyes
The formula of aluminium sulphide is al₂s₃. explain why the formula has a ratio of two aluminium ions for every three sulphide ions.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1
You know the right answer?
The formula of aluminium sulphide is al₂s₃. explain why the formula has a ratio of two aluminium ion...
Questions


Computers and Technology, 12.11.2019 20:31

History, 12.11.2019 20:31





Mathematics, 12.11.2019 20:31

Computers and Technology, 12.11.2019 20:31

Mathematics, 12.11.2019 20:31



Biology, 12.11.2019 20:31


English, 12.11.2019 20:31




Biology, 12.11.2019 20:31