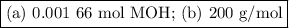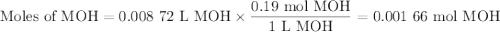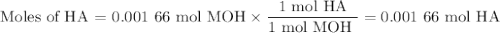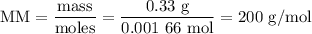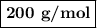Chemistry, 20.10.2019 06:50 goldenarrow
Using the concentration of the base and the volume of the base used, calculate the moles of the base used in the titration. then, using the mass of the acid, determine the molar mass of the acid.
data:
concentration of the base(naoh)= 0.19 m
volume of the base used= 8.72 ml
mass of the acid(unknown)= 0.33 g

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:00
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1

Chemistry, 22.06.2019 21:30
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3

Chemistry, 23.06.2019 01:00
What two factors can affect the properties of a hydrocarbon? a. the number of its carbon atoms and the number of single bonds b. the number of its carbon atoms and the arrangement of its atoms c. the arrangement of its atoms and the number of its double bonds
Answers: 1

Chemistry, 23.06.2019 02:50
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2
You know the right answer?
Using the concentration of the base and the volume of the base used, calculate the moles of the base...
Questions

Mathematics, 03.08.2019 21:30






History, 03.08.2019 21:30





Spanish, 03.08.2019 21:30

Mathematics, 03.08.2019 21:30

English, 03.08.2019 21:30



Health, 03.08.2019 21:30