Option (b) is the correct answer.
Explanation:
STP means standard temperature and pressure. At this condition pressure is 1 atm, volume is 22.4 L and temperature is 298 K.
Therefore, using ideal gas equation we will calculate the number of moles as follows.
PV = nRT
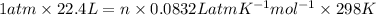
n = 0.903 mol
= 1 mol (approx)
Hence, it is known that molar mass of hydrogen molecule is 2 g/mol. Therefore, calculate mass of 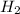 at STP as follows.
at STP as follows.
Number of moles = 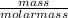
1 mol = 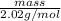
mass = 2.02 g
Thus, we can conclude that the mass of 22.4 L of H2 at STP is 2.02 grams.
























