Chemistry, 25.09.2019 21:30 taytaycola223
Giving bunch of points!
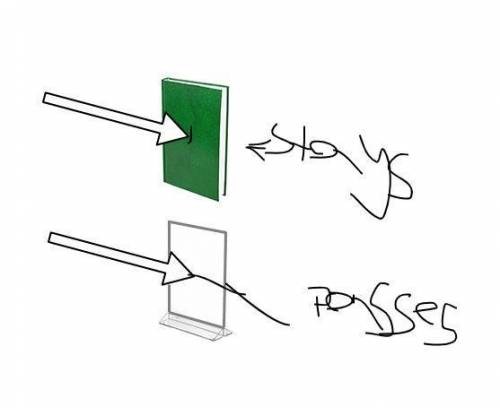
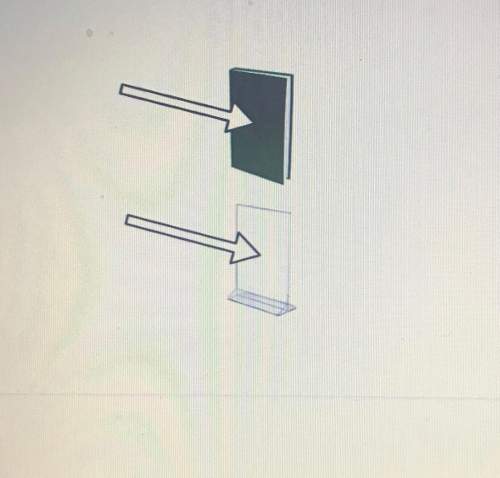
draw the rays, and insert text boxes to describe reflection, absorption, and transmission for the book and the glass.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
How many atoms are in 1.4 mil of phosphorus trifluoride (pf3)
Answers: 3

Chemistry, 22.06.2019 02:40
Achange in the number of neutrons in an atom will change an blank . when the number of protons changes in an atom, a new element will form.
Answers: 2

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
Giving bunch of points!
draw the rays, and insert text boxes to describe reflection, absorpt...
draw the rays, and insert text boxes to describe reflection, absorpt...
Questions



Mathematics, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01







Mathematics, 16.10.2020 08:01

Chemistry, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01


Mathematics, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01

Mathematics, 16.10.2020 08:01



Spanish, 16.10.2020 08:01