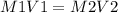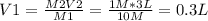Will give 50 points !
a 10 m concentrated stock solution of nacl is used to prepare 3 liter...

Will give 50 points !
a 10 m concentrated stock solution of nacl is used to prepare 3 liters of diluted 1 m solution. which of the following is true for the process used to achieve the required dilution?
the volume of stock solution used is less than 0.2 liters.
the volume of stock solution used is less than 3 liters. (i choose this answer)
the volume of the solvent used is less than 0.2 liters.
the volume of the solvent used is more than 3 liters.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 14:30
Select the word from the list that best fits the definition the nuclear family into which a person is born or adopted.
Answers: 2

Chemistry, 22.06.2019 17:00
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2

Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3
You know the right answer?
Questions

English, 08.10.2019 13:50





Mathematics, 08.10.2019 13:50

History, 08.10.2019 13:50


Physics, 08.10.2019 13:50

English, 08.10.2019 13:50

Mathematics, 08.10.2019 13:50


Health, 08.10.2019 13:50





Mathematics, 08.10.2019 14:00

Biology, 08.10.2019 14:00