Chemistry, 05.01.2020 14:31 twirlergirl800
Achemistry student weighs out 0.0634g of formic acid hcho2 into a 250.ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1800m naoh solution. calculate the volume of naoh solution the student will need to add to reach the equivalence point. be sure your answer has the correct number of significant digits.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 22.06.2019 18:40
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1
You know the right answer?
Achemistry student weighs out 0.0634g of formic acid hcho2 into a 250.ml volumetric flask and dilute...
Questions

Physics, 10.07.2019 17:20



Mathematics, 10.07.2019 17:20




Mathematics, 10.07.2019 17:20



Mathematics, 10.07.2019 17:30


Mathematics, 10.07.2019 17:30

Mathematics, 10.07.2019 17:30


SAT, 10.07.2019 17:30



Mathematics, 10.07.2019 17:30

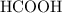 :
: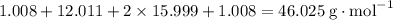 .
.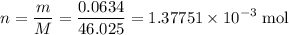 .
.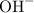 ion to neutralize each carbonyl group
ion to neutralize each carbonyl group 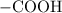 .
.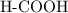 molecule. Each formula unit of NaOH supplies one
molecule. Each formula unit of NaOH supplies one 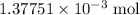 of formic acid in the volumetric flask. It will take the same number of NaOH formula units to reach the equivalence point.
of formic acid in the volumetric flask. It will take the same number of NaOH formula units to reach the equivalence point.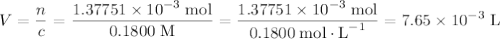 .
.