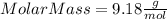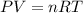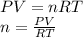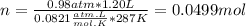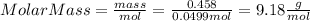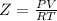Part 1. determine the molar mass of a 0.458-gram sample of gas having a volume of 1.20 l at 287 k and 0.980 atm. show your work. part 2. if this sample was placed under extreme pressure, describe how the actual volume would compare to the predicted volume. explain your answer.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1

Chemistry, 22.06.2019 17:30
Energy defines the different "states" of matter. in no more than 3 sentences, describe the amount of kinetic energy that each of the 3 states of matter possesses and relate that to the atom/molecular motion of each "state".
Answers: 2

You know the right answer?
Part 1. determine the molar mass of a 0.458-gram sample of gas having a volume of 1.20 l at 287 k an...
Questions

Computers and Technology, 29.08.2019 22:00

Mathematics, 29.08.2019 22:00


Mathematics, 29.08.2019 22:00




Biology, 29.08.2019 22:00


English, 29.08.2019 22:00


Geography, 29.08.2019 22:00

Mathematics, 29.08.2019 22:00



Mathematics, 29.08.2019 22:00

English, 29.08.2019 22:00



Biology, 29.08.2019 22:00