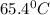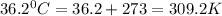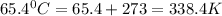Chemistry, 06.10.2019 09:10 yourfreeshoppep3u91x
At which of the following temperatures is the average kinetic energy of the molecules of a substance the greatest?
( a. 18.1 °c
( b. 20.4 °c
( c. 36.2 °c
( d. 65.4 °c

Answers: 2
Another question on Chemistry


Chemistry, 23.06.2019 00:30
An ice cube with a volume of 45.0ml and a density of 0.9000g/cm3 floats in a liquid with a density of 1.36g/ml. what volume of the cube is submerged in the liquid
Answers: 3

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2

Chemistry, 23.06.2019 05:00
1. true or false: minerals are inorganic. true false 2. inorganic means that something has never been found alive 3. halite is another name for and is a mineral with a cubic crystal pattern. table salt rock salt
Answers: 2
You know the right answer?
At which of the following temperatures is the average kinetic energy of the molecules of a substance...
Questions


Mathematics, 11.02.2021 01:30

English, 11.02.2021 01:30

World Languages, 11.02.2021 01:30

Mathematics, 11.02.2021 01:30

Mathematics, 11.02.2021 01:30


History, 11.02.2021 01:30










English, 11.02.2021 01:30


Biology, 11.02.2021 01:30