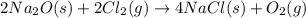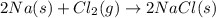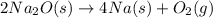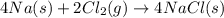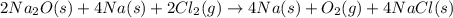Chemistry, 14.10.2019 08:00 mariap3504
In the final chemical equation, nacl and o2 are the products that are formed through the reaction between na2o and cl2. before you can add these intermediate chemical equations, you need to alter them by


Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 23.06.2019 00:20
What type of context clue you understand the meaning of quandary?
Answers: 3
You know the right answer?
In the final chemical equation, nacl and o2 are the products that are formed through the reaction be...
Questions


Health, 24.06.2021 05:10

English, 24.06.2021 05:10


Mathematics, 24.06.2021 05:10



Mathematics, 24.06.2021 05:10

Physics, 24.06.2021 05:10

Mathematics, 24.06.2021 05:10

Mathematics, 24.06.2021 05:10

Biology, 24.06.2021 05:10

History, 24.06.2021 05:10

Mathematics, 24.06.2021 05:10


Computers and Technology, 24.06.2021 05:10



Mathematics, 24.06.2021 05:10

Mathematics, 24.06.2021 05:10