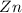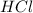When zinc reacts with hydrochloric acid, it produces hydrogen gas. as the reaction proceeds, why does the rate of production of hydrogen gas decrease? the concentration of hydrogen gas decreases. the concentration of the reactants decreases. the hydrogen gas formed inhibits the reaction. the hydrogen gas also reacts with the zinc metal.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1


Chemistry, 23.06.2019 03:50
What is the equation fort the alkaline zinc/manganese dioxide cell. a) anode b)cathode c)overall equations.
Answers: 2
You know the right answer?
When zinc reacts with hydrochloric acid, it produces hydrogen gas. as the reaction proceeds, why doe...
Questions

Physics, 05.10.2019 12:10





History, 05.10.2019 12:10

Physics, 05.10.2019 12:10

Mathematics, 05.10.2019 12:10

Mathematics, 05.10.2019 12:10

Mathematics, 05.10.2019 12:10

Mathematics, 05.10.2019 12:10

History, 05.10.2019 12:10

Social Studies, 05.10.2019 12:10


Mathematics, 05.10.2019 12:10

Social Studies, 05.10.2019 12:10


Computers and Technology, 05.10.2019 12:10

History, 05.10.2019 12:10

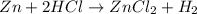
![Rate=k[A]^x[B]^y](/tpl/images/0383/1895/ddde1.png)