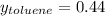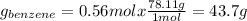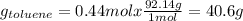Chemistry, 14.01.2020 15:31 citlalli30
Asolution of benzene (c6h6) and toluene (c7h8) is 25.0% benzene by mass. the vapor pressures of pure benzene and pure toluene at 25°c are 94.2 torr and 28.4 torr, respectively. assuming ideal behavior, calculate the following: (a) the vapor pressure of each solution component in the mixture (b) the total pressure above the solution (c) the composition of the vapor in mass percent why is the composition of the vapor different from the composition of the solution?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 12:00
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1

Chemistry, 22.06.2019 17:00
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1
You know the right answer?
Asolution of benzene (c6h6) and toluene (c7h8) is 25.0% benzene by mass. the vapor pressures of pure...
Questions


History, 29.08.2019 18:30

English, 29.08.2019 18:30






Mathematics, 29.08.2019 18:30


Computers and Technology, 29.08.2019 18:30



Mathematics, 29.08.2019 18:30

Mathematics, 29.08.2019 18:30

Chemistry, 29.08.2019 18:30


Mathematics, 29.08.2019 18:30

Computers and Technology, 29.08.2019 18:30

Chemistry, 29.08.2019 18:30

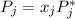
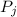 is the partial pressure of component j in the gas mixture above the solution,
is the partial pressure of component j in the gas mixture above the solution, 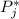 is the vapor pressure of the pure component and
is the vapor pressure of the pure component and 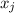 is the molar fraction of component j in the liquid mixture (in the solution).
is the molar fraction of component j in the liquid mixture (in the solution).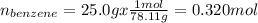
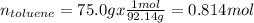
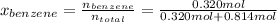 →
→ 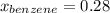
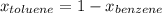 →
→ 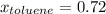
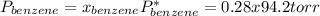
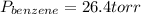
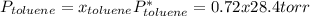
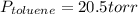
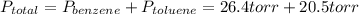
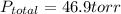
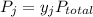
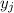 is the mole fraction of the gas j in the gas mixture and
is the mole fraction of the gas j in the gas mixture and 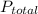 is the total pressure.
is the total pressure.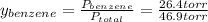
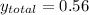
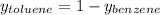 →
→