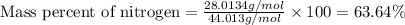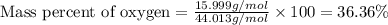What is the percentage composition of each element in dinitrogen monoxide, n2o?
a. 58.3...

Chemistry, 27.12.2019 21:31 jaiyaharoldjh
What is the percentage composition of each element in dinitrogen monoxide, n2o?
a. 58.32% n; 41.68% o
b. 60.55% n; 39.45% o
c. 63.64% n; 36.36% o
d. 62.66% n; 37.34% o

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3
You know the right answer?
Questions



Computers and Technology, 21.01.2021 22:50









Computers and Technology, 21.01.2021 22:50





Mathematics, 21.01.2021 22:50

History, 21.01.2021 22:50


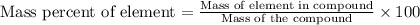
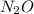 = 44.013 g/mol
= 44.013 g/mol![[2\times 14.0067]=28.0134g/mol](/tpl/images/0434/9605/d0294.png)