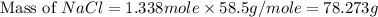Chemistry, 31.01.2020 23:04 sjjarvis53211
Part 1. a chemist reacted 15.0 liters of f2 gas with nacl in the laboratory to form cl2 and naf. use the ideal gas law equation to determine the mass of nacl that reacted with f2 at 280. k and 1.50 atm.
f2 + 2nacl → cl2 + 2naf
part 2. explain how you would determine the mass of sodium chloride that can react with the same volume of fluorine gas at stp.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 07:50
In which situation can a mixture always be called a solution
Answers: 3


Chemistry, 22.06.2019 23:30
What are the similarities between compounds and mixtures?
Answers: 3
You know the right answer?
Part 1. a chemist reacted 15.0 liters of f2 gas with nacl in the laboratory to form cl2 and naf. use...
Questions

English, 14.05.2021 22:00

Chemistry, 14.05.2021 22:00

English, 14.05.2021 22:00

Mathematics, 14.05.2021 22:00

Mathematics, 14.05.2021 22:00


Mathematics, 14.05.2021 22:00



Mathematics, 14.05.2021 22:00

Geography, 14.05.2021 22:00

English, 14.05.2021 22:00


Mathematics, 14.05.2021 22:00

Mathematics, 14.05.2021 22:00





 gas by using ideal gas equation.
gas by using ideal gas equation.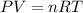
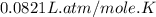
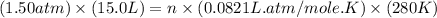
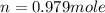
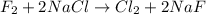
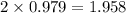 moles of NaCl
moles of NaCl
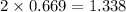 moles of NaCl
moles of NaCl