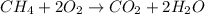Examine the unbalanced equation.
ch4 + o2 → co2 + h2o
what is the best classification for the unbalanced equation's reaction, and why?
it is a combustion reaction, because carbon dioxide (co2) is emitted as a product.
it is a combustion reaction, because oxygen (o2) reacts with a substance.
it is a synthesis reaction, because o2 combines with ch4 to create a new substance.
it is a synthesis reaction, because o2 displaces carbon (c) and hydrogen (h).

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 23.06.2019 02:00
Scientists are often interested in knowing the molar heat of combustion – the heat released during the combustion of one mole of a substance. use the periodic table to find molar masses. how many moles of ethanol are present in the sample?
Answers: 2
You know the right answer?
Examine the unbalanced equation.
ch4 + o2 → co2 + h2o
what is the best class...
ch4 + o2 → co2 + h2o
what is the best class...
Questions



Mathematics, 18.11.2019 14:31



Spanish, 18.11.2019 14:31

Geography, 18.11.2019 14:31

Chemistry, 18.11.2019 14:31


Physics, 18.11.2019 14:31




Chemistry, 18.11.2019 14:31


Social Studies, 18.11.2019 14:31


English, 18.11.2019 14:31


History, 18.11.2019 14:31