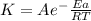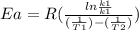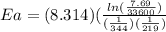Areaction is followed and found to have a rate constant of 3.36 × 104 m-1s-1 at 344 k and a rate constant of 7.69 m-1s-1 at 219 k. determine the activation energy for this reaction. a reaction is followed and found to have a rate constant of 3.36 × 104 m-1s-1 at 344 k and a rate constant of 7.69 m-1s-1 at 219 k. determine the activation energy for this reaction. 12.5 kj/mol 11.5 kj/mol 23.8 kj/mol 58.2 kj/mol 42.0 kj/mol

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3

Chemistry, 22.06.2019 10:00
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1


Chemistry, 22.06.2019 23:10
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 3
You know the right answer?
Areaction is followed and found to have a rate constant of 3.36 × 104 m-1s-1 at 344 k and a rate con...
Questions


Mathematics, 17.10.2019 20:00

Geography, 17.10.2019 20:00


English, 17.10.2019 20:00

History, 17.10.2019 20:00




Mathematics, 17.10.2019 20:00



Biology, 17.10.2019 20:00

Computers and Technology, 17.10.2019 20:00


Geography, 17.10.2019 20:00

Biology, 17.10.2019 20:00

Mathematics, 17.10.2019 20:00