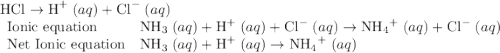Chemistry, 18.12.2019 10:31 lezapancakes13
Which of the chemical equations below are acid-base (proton transfer) reactions? select all that apply. mg(oh)2 (s) + h2so4 (aq) → mgso4 (s) + 2 h2o (l) fe(no3)3 (aq) + 3 koh (aq) → fe(oh)3 (s) + 3 kno3 (aq) zn (s) + cu(no3)2 (aq) → zn(no3)2 (aq) + cu (s) mg (s) + cu(no3)2 (aq) --> mg(no3)3 (aq) + cu (s) hcl (aq) + koh (aq) → kcl (aq) + h2o (l) nh3 (aq) + hcl (aq) → nh4cl (aq)?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1


Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
Which of the chemical equations below are acid-base (proton transfer) reactions? select all that ap...
Questions

Arts, 29.07.2019 23:00

Mathematics, 29.07.2019 23:00


Biology, 29.07.2019 23:00

History, 29.07.2019 23:00

History, 29.07.2019 23:00


Mathematics, 29.07.2019 23:00



History, 29.07.2019 23:00


Advanced Placement (AP), 29.07.2019 23:00

Biology, 29.07.2019 23:00