
When magnesium burns in air, it combines with oxygen to form magnesium oxide according to the following equation. what mass in grams of magnesium oxide is produced from 2.00 mol of magnesium?
2mg(s) + o2(g) -> 2mgo(s)
what is the given quantity?
what is the unknown quantity?
write the two conversion factors needed to solve the problem.
calculate the molar mass of the unknown using the given quantity and the two conversion factors.

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 23:00
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2

Chemistry, 23.06.2019 03:50
How many moles of potassium are needed to react completely with 12.8 moles of magnessium bromide?
Answers: 2

Chemistry, 23.06.2019 08:00
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 1
You know the right answer?
When magnesium burns in air, it combines with oxygen to form magnesium oxide according to the follow...
Questions


Arts, 12.07.2019 06:00


Mathematics, 12.07.2019 06:00

Mathematics, 12.07.2019 06:00







Mathematics, 12.07.2019 06:00



Social Studies, 12.07.2019 06:00

Mathematics, 12.07.2019 06:00

Mathematics, 12.07.2019 06:00

Mathematics, 12.07.2019 06:00


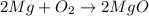
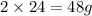 of magnesium produce=
of magnesium produce=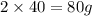 of MgO.
of MgO.


