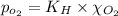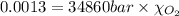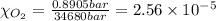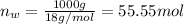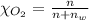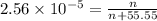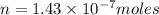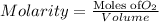Chemistry, 03.10.2019 05:50 yyyyyyyyy8938
Using henry's law, calculate the molar concentration of o2 in the surface water of a mountain lake saturated with air at 20 ∘c and an atmospheric pressure of 685 torr .

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:30
Strong conductivity of plasma allows it to act and react as and
Answers: 2

Chemistry, 22.06.2019 12:40
In the following table, all the columns for the element calcium are filled out correctly. element electron structure of atom electron structure of ion net ionic charge calcium 1s22s22p63s23p64s2 1s32s22p63s23p64s1 +1 true false
Answers: 2

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
Using henry's law, calculate the molar concentration of o2 in the surface water of a mountain lake s...
Questions



Chemistry, 29.08.2019 09:00

Health, 29.08.2019 09:00


Chemistry, 29.08.2019 09:00

History, 29.08.2019 09:00

Health, 29.08.2019 09:00



Biology, 29.08.2019 09:00



History, 29.08.2019 09:00

English, 29.08.2019 09:00

Mathematics, 29.08.2019 09:00

Computers and Technology, 29.08.2019 09:00

Social Studies, 29.08.2019 09:00

Mathematics, 29.08.2019 09:00

Mathematics, 29.08.2019 09:00

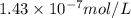 .
.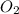 gas = 685 torr = 0.8905 bar
gas = 685 torr = 0.8905 bar