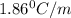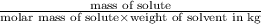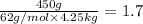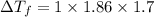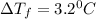Ethylene glycol (molar mass = 62 g/mol)is used as an antifreeze in cars. if 450 g of ethylene glycol is added to 4.25 kg of water, what is the molality? calculate how much the freezing point of water will be lowered. the freezing point depression constant for water is kf = -1.86°c/m. show your work.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 22.06.2019 19:30
Anurse used a 0.02-mg/l solution of disinfection to clean a patients wound. what is the concentration of the solution expressed as a percentage?
Answers: 1

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1
You know the right answer?
Ethylene glycol (molar mass = 62 g/mol)is used as an antifreeze in cars. if 450 g of ethylene glycol...
Questions




Mathematics, 04.04.2020 12:03


Mathematics, 04.04.2020 12:03





Social Studies, 04.04.2020 12:04


History, 04.04.2020 12:04







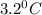
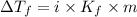
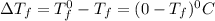 = Depression in freezing point
= Depression in freezing point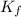 = freezing point constant =
= freezing point constant =