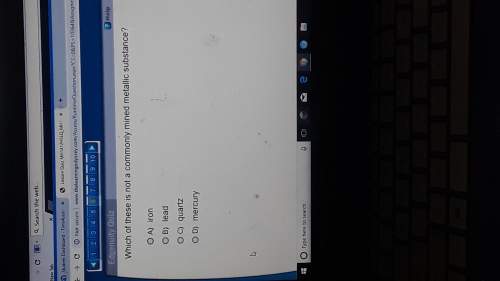
Answers: 1
Another question on Chemistry


Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2

Chemistry, 23.06.2019 09:10
Complete the following radioactive decay problem. tan+on-? c+th
Answers: 1

Chemistry, 23.06.2019 10:30
Fill in the blanks for the following statements: the rms speed of the molecules in a sample of h2 gas at 300 k will be times larger than the rms speed of o2 molecules at the same temperature, and the ratio µrms (h2) / µrms (o2) with increasing temperature. a not enough information is given to answer this question b sixteen, will not change c four, will not change d four, will increase e sixteen, will decrease
Answers: 2
You know the right answer?
How many moles are equivalent to 55.5g of nano3...
Questions


Mathematics, 22.04.2021 01:00

Social Studies, 22.04.2021 01:00

English, 22.04.2021 01:00

English, 22.04.2021 01:00


Biology, 22.04.2021 01:00

English, 22.04.2021 01:00



Mathematics, 22.04.2021 01:00

Mathematics, 22.04.2021 01:00

Mathematics, 22.04.2021 01:00

Mathematics, 22.04.2021 01:00





Mathematics, 22.04.2021 01:00

Mathematics, 22.04.2021 01:00