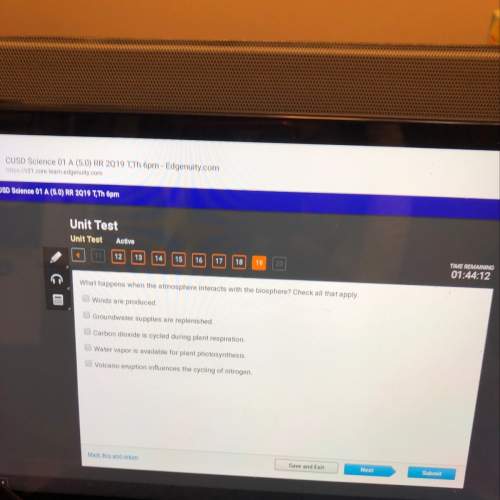
Chemistry, 05.10.2019 13:00 yuppcami9929
If you have 3.50 moles of hydrogen and 5.00moles of nitrogen to produce ammonia, witch element is the reactant in excess? 3h2+n2=2nh3

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 07:00
In the cathode ray tube experiment, j. j. thomson passed an electric current through different gases inside a cathode ray tube in the presence of an electric field. in which two ways did this experiment change scientists’ understanding of the atom?
Answers: 2

Chemistry, 22.06.2019 12:30
Which element has the lowest electronegativity? calcium(ca) gallium(ga) selenium(se) bromine(br)
Answers: 1
You know the right answer?
If you have 3.50 moles of hydrogen and 5.00moles of nitrogen to produce ammonia, witch element is th...
Questions



English, 13.02.2022 07:30



Social Studies, 13.02.2022 07:30




Mathematics, 13.02.2022 07:30

Mathematics, 13.02.2022 07:30



History, 13.02.2022 07:30




Mathematics, 13.02.2022 07:30

Mathematics, 13.02.2022 07:30

Chemistry, 13.02.2022 07:30