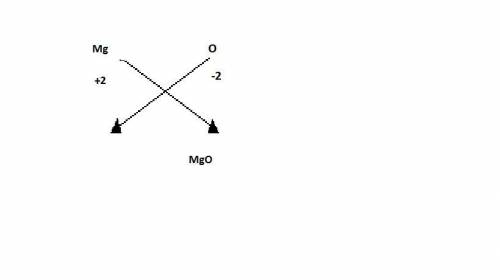
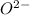Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2

Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2
You know the right answer?
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chem...
Questions

Biology, 06.07.2019 01:30



Biology, 06.07.2019 01:30

Chemistry, 06.07.2019 01:30


Mathematics, 06.07.2019 01:30



Geography, 06.07.2019 01:30

Mathematics, 06.07.2019 01:30

Chemistry, 06.07.2019 01:30





Geography, 06.07.2019 01:30

Mathematics, 06.07.2019 01:30


Mathematics, 06.07.2019 01:30

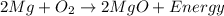
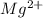 and oxygen gains two electrons to form
and oxygen gains two electrons to form