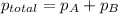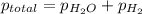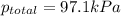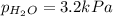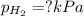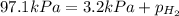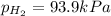Chemistry, 23.06.2019 10:30 lvoltin1073
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3 2 kilopascals, what is the partial pressure of the hydrogen?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:00
How is the shape of the poem “peer” connected to its meaning?
Answers: 2

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1


Chemistry, 23.06.2019 08:00
At 35.0°c and 3.00 atm pressure, a gas has a volume of 1.40 l. what pressure does the gas have at 0.00°c and a volume of 0.950 l? which equation should you use? p2= p1v1t2/t1v2what is the pressure of the gas? 3.92 atm these are the answers
Answers: 1
You know the right answer?
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water va...
Questions


Biology, 22.07.2019 11:00

Biology, 22.07.2019 11:00

Business, 22.07.2019 11:00






Geography, 22.07.2019 11:00



Geography, 22.07.2019 11:00





Mathematics, 22.07.2019 11:00