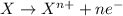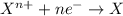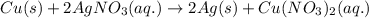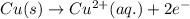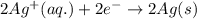Chemistry, 24.06.2019 13:00 melikefood01
Which of the following is an essential condition for a redox reaction? the reactants should be two elements. the reactants should be two compounds. the oxidation state of all the atoms should change. the oxidation state of at least two atoms should change.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
You know the right answer?
Which of the following is an essential condition for a redox reaction? the reactants should be two...
Questions


Mathematics, 17.09.2019 06:00


Social Studies, 17.09.2019 06:00

Computers and Technology, 17.09.2019 06:00

Physics, 17.09.2019 06:00


Mathematics, 17.09.2019 06:00





Mathematics, 17.09.2019 06:00


Mathematics, 17.09.2019 06:00


Chemistry, 17.09.2019 06:00



History, 17.09.2019 06:00