
What is the theoretical yield of copper produced by the reaction of: 0.5 g al reacted with 3.5 g cucl2 x h2o? (cucl2 x h2o is a dihydrate when calculating the molecular weight) hint.) first find the limiting reactant, then the molecular weights of copper by the aluminum and the (copper chloride times water).

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:20
Use the gizmo to find the concentration of the mystery ch3cooh. use the titrant and indicator shown below perform the titration. what is the titrant volume? titrant analyte indicator titrant volume analyte concentration naoh ch3cooh phenophthalein select one: a. 20.0 ml b. 27.0 ml c. 30.0 ml d. 24.0 ml
Answers: 2

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1

Chemistry, 23.06.2019 01:30
Which is an example of a highly unstable isotope that is often used in fission reactions?
Answers: 1

Chemistry, 23.06.2019 08:00
How many distinct monochlorinated products, including stereoisomers, can result when the alkane below is heated in the presence of cl2? 3 4 5 6 7?
Answers: 3
You know the right answer?
What is the theoretical yield of copper produced by the reaction of: 0.5 g al reacted with 3.5 g cu...
Questions







Mathematics, 08.05.2021 01:00


Mathematics, 08.05.2021 01:00

Mathematics, 08.05.2021 01:00



Arts, 08.05.2021 01:00

Mathematics, 08.05.2021 01:00

Chemistry, 08.05.2021 01:00





Physics, 08.05.2021 01:00

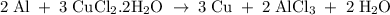
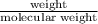
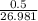
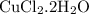 in reaction =
in reaction = 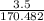
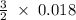
 170.482 grams of
170.482 grams of 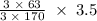 grams of Cu
grams of Cu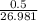 = 0.018 moles of Al.
= 0.018 moles of Al.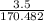 = 0.020 moles of CuCl₂.2H₂O.
= 0.020 moles of CuCl₂.2H₂O.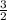 ×0.018 = 0.027.
×0.018 = 0.027.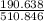 ×3.5 = 1.306g of Cu will produce.
×3.5 = 1.306g of Cu will produce.

