
Chemistry, 26.06.2019 22:00 ineedtopeebeforethec
Given the following chemical reaction: 2 o2 + ch4 co2 + 2 h2o what mass of co2 is produced in the reaction of 4 moles of ch4 in excess o2? 6 c 12.01 carbon 1 h 1.01 hydrogen 8 o 16.00 oxygen a. 44 grams b. 64 grams c. 164 grams d. 176 grams

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2


Chemistry, 22.06.2019 14:10
Aconcentrated solution of ammonia is 14.8m and has a density of 0.899g/l. what is the concentration of ammonia in this solution in weight percent (%w/w)?
Answers: 1

Chemistry, 23.06.2019 01:00
What is the chemical name of the compound ti2o3? use the list of polyatomic ions and the periodic table to you answer.
Answers: 1
You know the right answer?
Given the following chemical reaction: 2 o2 + ch4 co2 + 2 h2o what mass of co2 is produced in the r...
Questions

History, 29.01.2020 01:59

Biology, 29.01.2020 01:59


Health, 29.01.2020 01:59




Mathematics, 29.01.2020 01:59










Chemistry, 29.01.2020 02:00


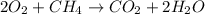
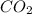
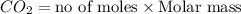
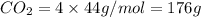
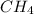 in excess
in excess 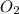 .
.


