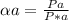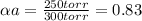Chemistry, 27.06.2019 14:30 JewelzSkullz
Substances a and b are both volatile liquids with p*a = 300 torr, p*b = 250 torr, and kb = 200 torr (concentration expressed in mole fraction). when xa = 0.9, bb = 2.22 mol kg−1, pa = 250 torr, and pb = 25 torr. calculate the activities and activity coefficients of a and b. use the mole fraction, raoult's law basis system for a and the henry's law basis system (both mole fractions and molalities) for b.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1
You know the right answer?
Substances a and b are both volatile liquids with p*a = 300 torr, p*b = 250 torr, and kb = 200 torr...
Questions

Mathematics, 22.10.2020 06:01


Mathematics, 22.10.2020 06:01



Mathematics, 22.10.2020 06:01

Mathematics, 22.10.2020 06:01

Mathematics, 22.10.2020 06:01


Mathematics, 22.10.2020 06:01

Mathematics, 22.10.2020 06:01

Social Studies, 22.10.2020 06:01

Mathematics, 22.10.2020 06:01

Chemistry, 22.10.2020 06:01

Mathematics, 22.10.2020 06:01

Mathematics, 22.10.2020 06:01

English, 22.10.2020 06:01

Mathematics, 22.10.2020 06:01

History, 22.10.2020 06:01