
Chemistry, 27.06.2019 19:30 ccarwile01
Determine the rate law, including the values of the orders and rate law constant, for the following reaction using the experimental data provided. (4 points) a + b yieldsproducts trial [a] [b] rate 1 0.10 m 0.20 m 1.2 × 10-2 m/min 2 0.10 m 0.40 m 4.8 × 10-2 m/min 3 0.20 m 0.40 m 9.6 × 10-2 m/min

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3


Chemistry, 23.06.2019 14:40
Uuestons niuthe no. of millimoles of hcl required to neutralize 10 ml of 0.2 m na2co3 is(a) 2.0 m mole(b) 4.0 m mole(c) 0.2 m mole(d) 0.4 m mole
Answers: 1

Chemistry, 23.06.2019 15:30
If the theoretical yield of a reaction is 26.0 grams and you actually recovered 22.0 grams, what is the percent yield?
Answers: 2
You know the right answer?
Determine the rate law, including the values of the orders and rate law constant, for the following...
Questions


Mathematics, 07.10.2019 20:30

Mathematics, 07.10.2019 20:30

Geography, 07.10.2019 20:30

Mathematics, 07.10.2019 20:30

Mathematics, 07.10.2019 20:30

English, 07.10.2019 20:30


Mathematics, 07.10.2019 20:30





Health, 07.10.2019 20:30

English, 07.10.2019 20:30


Mathematics, 07.10.2019 20:30

Mathematics, 07.10.2019 20:30

Social Studies, 07.10.2019 20:30

Social Studies, 07.10.2019 20:30

![k[A]^1[B]^2](/tpl/images/0024/4376/f6c70.png) , order with respect to A is 1, order with respect to B is 2 and total order is 3. Rate law constant is
, order with respect to A is 1, order with respect to B is 2 and total order is 3. Rate law constant is 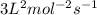
![Rate=k[A]^x[B]^y](/tpl/images/0024/4376/ddde1.png)
![1.2\times 10^{-2}=k[0.10]^x[0.20]^y](/tpl/images/0024/4376/c938a.png) (1)
(1)![4.8\times 10^{-2}=k[0.10]^x[0.40]^y](/tpl/images/0024/4376/a7ba9.png) (2)
(2)![\frac{4.8\times 10^{-2}}{1.2\times 10^{-2}}=\frac{k[0.10]^x[0.40]^y}{k[0.10]^x[0.20]^y}](/tpl/images/0024/4376/b4c1f.png)
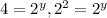 therefore y=2.
therefore y=2.![9.6\times 10^{-2}=k[0.20]^x[0.40]^y](/tpl/images/0024/4376/18d47.png) (4)
(4)![\frac{9.6\times 10^{-2}}{4.8\times 10^{-2}}=\frac{k[0.20]^x[0.40]^y}{k[0.10]^x[0.40]^y}](/tpl/images/0024/4376/61313.png)
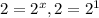 , x=1
, x=1![Rate=k[A]^1[B]^2](/tpl/images/0024/4376/ca297.png)
![1.2\times 10^{-2}=k[0.10]^1[0.20]^2](/tpl/images/0024/4376/f6a0d.png)
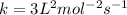 .
.

