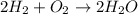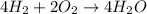Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3

Chemistry, 22.06.2019 10:40
If an area has high air pressure and low humidity, what type of weather will it most likely have? plz !
Answers: 1
You know the right answer?
How many moles of water are produced when 5 moles of hydrogen gas react with 2 moles of oxygen gas?...
Questions

Mathematics, 24.01.2020 15:31


History, 24.01.2020 15:31

Mathematics, 24.01.2020 15:31

Mathematics, 24.01.2020 15:31




Biology, 24.01.2020 15:31

Computers and Technology, 24.01.2020 15:31

Biology, 24.01.2020 15:31

English, 24.01.2020 15:31




Mathematics, 24.01.2020 15:31

History, 24.01.2020 15:31


Mathematics, 24.01.2020 15:31

Mathematics, 24.01.2020 15:31