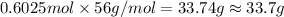And explain how to get the answer! 1. given the equation: 2na+cl2--> 2nacl if 200 grams of naci is produced, how many grams of na must be reacted with excess chlorine? a. 58.43g nab. 78.65g nac. 22.98g nad. 3.4g na2. given the equation: 2k+2h2o--> 2koh+h2 if 23.5 grams of potassium are reacted with excess water, how many grams of potassium hydroxide will be formed? a. 33.7g kohb. 56.08g kohc. 39.09g kohd. 17.99g koh

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 14:30
What state of matter is ice a. liquid b. element c. solid d. gas
Answers: 1

Chemistry, 23.06.2019 00:00
If many scientists conduct the same or similar experiments, and all obtain similar results, a can be written, which is a generally agreed-upon statement that explains and predicts how a natural phenomenon works.
Answers: 1

Chemistry, 23.06.2019 01:10
Can someone check my work 98 5.05 acids and bases for this assignment you will be comparing acids and bases. the chart below will you organize the information needed: acids bases chemical properties (2) deodorant detergent vinger dish soap physical properties (2) orange juice toilet cleaner drain cleaner window cleaner ph level acid ph goes from 0-4 bases ph goes from 10-14 examples around you (2) vinger coffee lemon juice dark chocolate
Answers: 3
You know the right answer?
And explain how to get the answer! 1. given the equation: 2na+cl2--> 2nacl if 200 grams of naci...
Questions



Mathematics, 11.10.2019 08:10



Mathematics, 11.10.2019 08:10

Mathematics, 11.10.2019 08:10


History, 11.10.2019 08:10

Law, 11.10.2019 08:10


Mathematics, 11.10.2019 08:20

Mathematics, 11.10.2019 08:20

Mathematics, 11.10.2019 08:20

History, 11.10.2019 08:20


English, 11.10.2019 08:20

Social Studies, 11.10.2019 08:20

Mathematics, 11.10.2019 08:20

Biology, 11.10.2019 08:20

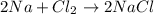
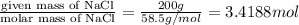
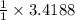 moles of Na.
moles of Na.
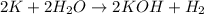
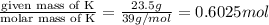
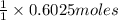 of KOH
of KOH