
Chemistry, 28.06.2019 03:00 jetblackcap
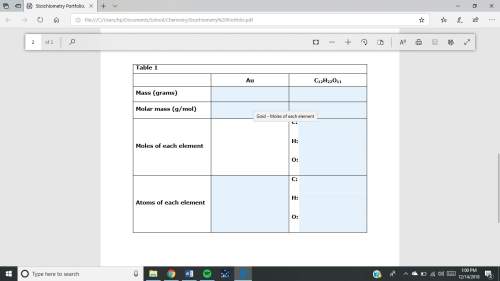
1. a sample of gold (au) has a mass of 35.12 g. a. calculate the number of moles of gold (au) in the sample and record in table 1. show your work. b. calculate the number of atoms of gold (au) in the sample and record in table 1. show your work. 2. a sample of table sugar (sucrose, c12h22o11) has a mass of 1.202 g. a. calculate the number of moles of c12h22o11 contained in the sample and record in table 1. show your work. b. calculate the moles of each element in c12h22o11 and record in table 1. show your work. c. calculate the number of atoms of each type in c12h22o11 and record in table 1. show your work. table looks like this: column 1 column 2 column 3 au c12h22o11mass (grams)molar mass (g/mol)moles of each element c: h: o: atoms of each element c: h: o: fast!

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:30
10. according to the law of conservation of mass, how does the mass of the products in a chemical reaction compare to the mass of the reactants?
Answers: 3

Chemistry, 21.06.2019 21:30
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1

Chemistry, 22.06.2019 21:30
How can the periodic table be used to predict the behavior of elements?
Answers: 1

You know the right answer?
1. a sample of gold (au) has a mass of 35.12 g. a. calculate the number of moles of gold (au) in the...
Questions

Mathematics, 04.05.2021 15:20

Health, 04.05.2021 15:20

Chemistry, 04.05.2021 15:30

Computers and Technology, 04.05.2021 15:30

Computers and Technology, 04.05.2021 15:30

Biology, 04.05.2021 15:30




Physics, 04.05.2021 15:30

Mathematics, 04.05.2021 15:30











