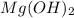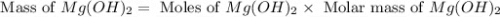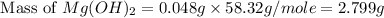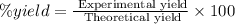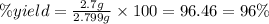Select the correct answer. excess sodium hydroxide is added to a solution containing 4.6 grams of magnesium chloride. a reaction takes place according to this equation: 2naoh(aq) + mgcl2(aq) → 2nacl(aq) + mg(oh)2(s). the magnesium hydroxide produced by the reaction was collected and weighed. if the mass of the magnesium hydroxide was 2.7 grams, what was the percent yield? use the periodic table. a. 48% b. 59% c. 61% d. 96%

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 15:30
If 200.0g of copper(ll) sulfate react with an excess of zinc metal, what is the theoretical yield of copper
Answers: 1


Chemistry, 21.06.2019 23:00
Layers of rock containing fossils, like the layers illustrated here, are most likely composed of rocks.
Answers: 2

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2
You know the right answer?
Select the correct answer. excess sodium hydroxide is added to a solution containing 4.6 grams of ma...
Questions

English, 28.01.2020 08:31



Mathematics, 28.01.2020 08:31

Chemistry, 28.01.2020 08:31





Mathematics, 28.01.2020 08:31

Biology, 28.01.2020 08:31

Mathematics, 28.01.2020 08:31

Mathematics, 28.01.2020 08:31

Mathematics, 28.01.2020 08:31



Mathematics, 28.01.2020 08:31

History, 28.01.2020 08:31


World Languages, 28.01.2020 08:31

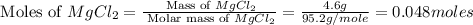
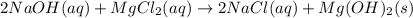
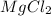 react to give 1 mole of
react to give 1 mole of