

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

Chemistry, 22.06.2019 21:30
Which of the following changes will decrease the total amount of gaseous solute able to be dissolved in a liter of liquid water? (2 points) decreasing temperature decreasing pressure decreasing surface area decreasing solute concentration
Answers: 1

You know the right answer?
Need answer soon plz ! if 8 mol of nh3 reacts with 14 mol of o2, how many moles of h2o will be prod...
Questions






Mathematics, 31.05.2021 17:40

Computers and Technology, 31.05.2021 17:40


Health, 31.05.2021 17:40



Mathematics, 31.05.2021 17:40

History, 31.05.2021 17:40



Mathematics, 31.05.2021 17:40



English, 31.05.2021 17:40

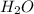 will be produced.
will be produced.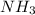 = 8 moles
= 8 moles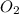 = 14 moles
= 14 moles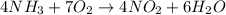
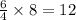 moles of
moles of 

