
Chemistry, 29.06.2019 10:30 ilovebeanieboos
If lithium loses an electron to become li+ what is the average atomic mass of the lithium ion? explain how you arrived at your answer

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2



Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
You know the right answer?
If lithium loses an electron to become li+ what is the average atomic mass of the lithium ion? expl...
Questions




Mathematics, 13.07.2019 12:00

Business, 13.07.2019 12:00



Mathematics, 13.07.2019 12:00

Biology, 13.07.2019 12:00




Mathematics, 13.07.2019 12:00

Mathematics, 13.07.2019 12:00

Health, 13.07.2019 12:00

Mathematics, 13.07.2019 12:00



Mathematics, 13.07.2019 12:00

Social Studies, 13.07.2019 12:00

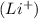 , the average atomic mass of the ion remains the same as the neutral atom.
, the average atomic mass of the ion remains the same as the neutral atom.

