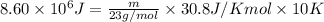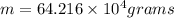Liquid sodium is being considered as an engine coolant. how many grams of liquid sodium (minimum) are needed to absorb 8.60 mj of energy (in the form of heat) if the temperature of the sodium is not to increase by more than 10.0 °c? use cp= 30.8 j/(k·mol) for na(l) at 500 k.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1

Chemistry, 23.06.2019 11:30
Bridget is in science class. her teacher gives her two unknown substances and asks her to determine their relative ph. she places a piece of red litmus paper into both substances. the litmus paper turns purple when she places it into substance i. the litmus paper turns blue when she places it into substance ii. a. substance i is a neutral substance and substance ii is an acid. b. substance i is a neutral substance and substance ii is a base. c. substance i is an acid and substance ii is a base. d. substance i is a base and substance ii is a neutral substance.
Answers: 1
You know the right answer?
Liquid sodium is being considered as an engine coolant. how many grams of liquid sodium (minimum) ar...
Questions

Mathematics, 10.11.2021 19:30


Biology, 10.11.2021 19:30


Chemistry, 10.11.2021 19:30




Social Studies, 10.11.2021 19:30

History, 10.11.2021 19:30


Geography, 10.11.2021 19:30

Mathematics, 10.11.2021 19:30


English, 10.11.2021 19:30

Mathematics, 10.11.2021 19:30

Health, 10.11.2021 19:30


Mathematics, 10.11.2021 19:30

Mathematics, 10.11.2021 19:30

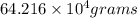 of liquid sodium is required.
of liquid sodium is required.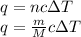
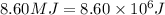 (Conversion factor: 1MJ = 1000000J)
(Conversion factor: 1MJ = 1000000J)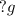
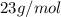
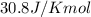
 = change in temperature,
= change in temperature, 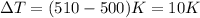 (Conversion factor: 0°C = 273K)
(Conversion factor: 0°C = 273K)