Chemistry, 29.06.2019 22:30 annyarias1036
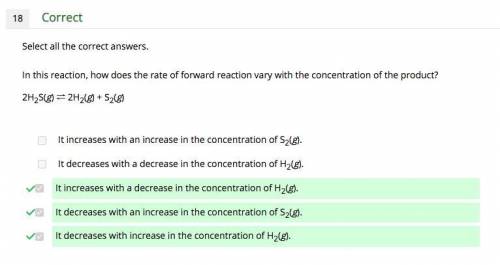
In this reaction, how does the rate of forward reaction vary with the concentration of the product? 2h2s(g) ⇌ 2h2(g) + s2(g) a. it increases with an increase in the concentration of s2(g). b. it decreases with a decrease in the concentration of h2(g). c. it increases with a decrease in the concentration of h2(g). d. it decreases with an increase in the concentration of s2(g). e. it decreases with increase in the concentration of h2(g). can choose more than one

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
During chemistry class, carl performed several lab tests on two white solids. the results of three tests are seen in the data table. based on this data, carl has concluded that substance b must have bonds.
Answers: 2

Chemistry, 22.06.2019 06:10
56.16 gregor mendel was the first scientist to use statistics to analyze scientific data. before mendel's experiments, scientists believed that organisms acquired traits from their environment and passed them on to their offspring. after mendel's discoveries were accepted, scientists realized that traits passed to offspring were the result of genes being passed from parents to offspring. this is an example of pls
Answers: 1

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1
You know the right answer?
In this reaction, how does the rate of forward reaction vary with the concentration of the product?...
Questions



Mathematics, 19.01.2022 19:30


SAT, 19.01.2022 19:30



Social Studies, 19.01.2022 19:30


Computers and Technology, 19.01.2022 19:30

Mathematics, 19.01.2022 19:30


Social Studies, 19.01.2022 19:40

Social Studies, 19.01.2022 19:40