
Chemistry, 30.06.2019 04:00 gracie6313
Based on reference table s, atoms of which of these elements have the strongest attraction for the electrons in a chemical bond? a) si b) p c) al d) s

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:40
Consider the nuclear equation below. 239/94 pu—-> x+ 4/2 he. what is x?
Answers: 2

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3

Chemistry, 23.06.2019 02:10
Detrimental the length of the object shown 1. 97.8mm 2. 97.80 mm 3. 97mm 4. 98mm
Answers: 2

Chemistry, 23.06.2019 03:40
Write the overall equation for the reaction occurring in lithium battery?
Answers: 3
You know the right answer?
Based on reference table s, atoms of which of these elements have the strongest attraction for the e...
Questions










Mathematics, 12.08.2020 21:01










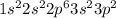 .
.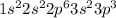 .
.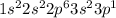 .
.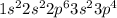 .
. 

