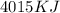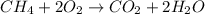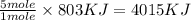Chemistry, 01.07.2019 00:30 smariedegray
The combustion of 1 mol ch4 releases 803 kj of energy. how much energy is produced from the combustion of 5.00 mol ch4.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3


Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
You know the right answer?
The combustion of 1 mol ch4 releases 803 kj of energy. how much energy is produced from the combusti...
Questions


Mathematics, 20.01.2021 07:10

Mathematics, 20.01.2021 07:10

Social Studies, 20.01.2021 07:10

English, 20.01.2021 07:10

English, 20.01.2021 07:10

Business, 20.01.2021 07:10

Health, 20.01.2021 07:10


Mathematics, 20.01.2021 07:20


History, 20.01.2021 07:20

Mathematics, 20.01.2021 07:20



Social Studies, 20.01.2021 07:20

Mathematics, 20.01.2021 07:20

English, 20.01.2021 07:20


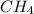 is
is