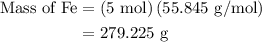Chemistry, 01.07.2019 13:00 amycressey1970
The following data was collected when a reaction was performed experimentally in the laboratory. fe2o3 al al2o3 fe starting amount in reaction 3 moles 5 moles ? ? determine the maximum amount of fe that was produced during the experiment. explain how you determined this amount.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 19:00
Avolleyball player hit a ball with a mass of 0.25 kg. the average acceleration of the ball is 15.5 m/s². how much force did the volleyball player apply to the ball? 62.0 n 3.87 n 62.0 m/s² 3.87 m/s²
Answers: 2

Chemistry, 23.06.2019 00:30
If there are 3.5 moles of koh, how many moles of naoh can be produced? question 1 options: a)3.0 moles naoh b)3.5 moles naoh c)1 moles naoh d)9 moles naoh
Answers: 1

Chemistry, 23.06.2019 05:00
Select the statement that describe chemical properties a. antacid tablets neutralize stomach acid b. helium is the lightest monatomic element c. water freezes at 0 celsius d. mercury is liquid at room temperature
Answers: 3
You know the right answer?
The following data was collected when a reaction was performed experimentally in the laboratory. fe2...
Questions









English, 04.07.2019 11:00





Biology, 04.07.2019 11:00




English, 04.07.2019 11:00


English, 04.07.2019 11:00

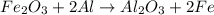
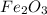 = 3 moles
= 3 moles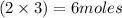 of Al
of Al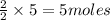 of Fe.
of Fe.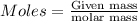
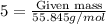
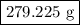 of Fe is produced during the given experiment.
of Fe is produced during the given experiment.
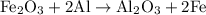
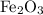 reacts with two moles of Al to produce one mole of
reacts with two moles of Al to produce one mole of 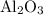 and two moles of Fe.
and two moles of Fe.
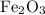 and 5 moles of Al. Therefore the amount of Fe produced by 3 moles of
and 5 moles of Al. Therefore the amount of Fe produced by 3 moles of 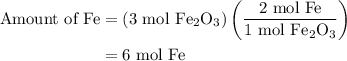
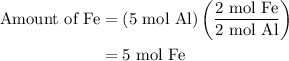
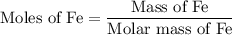 ...... (1)
...... (1) 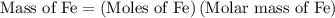 ...... (2)
...... (2)