
Chemistry, 02.07.2019 01:00 pattydixon6
An unbalanced chemical equation is shown: h2o2 → 2h2o + o2 which of the following statements explains why the equation is not balanced? a two h2o2 molecules should decompose to form the given products. b four h2o2 molecules should decompose to form the given products. c two h2o2 molecules should undergo a synthesis reaction to form the given products. d four h2o2 molecules should undergo a synthesis reaction to form the given products.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 10:40
Asolid that forms and separates from a liquid mixture is called
Answers: 2

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3
You know the right answer?
An unbalanced chemical equation is shown: h2o2 → 2h2o + o2 which of the following statements explai...
Questions





Mathematics, 12.11.2019 11:31







Chemistry, 12.11.2019 11:31

Mathematics, 12.11.2019 11:31

Mathematics, 12.11.2019 11:31


Advanced Placement (AP), 12.11.2019 11:31

Chemistry, 12.11.2019 11:31

Mathematics, 12.11.2019 11:31


English, 12.11.2019 11:31

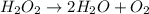

 molecules should decompose to form the given products."
molecules should decompose to form the given products."

