
Chemistry, 02.07.2019 15:30 Jordan0423
How many moles of potassium chloride and oxygen can be produced from 100.0g of potassium chlorate? the balanced equation is: 2 kclo3=2kcl+3o2

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:00
For each of the following types of reactions, write a general reaction formula in the symbolic form—for example, a + b → ab. single-displacement double-displacement synthesis decomposition
Answers: 1

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2
You know the right answer?
How many moles of potassium chloride and oxygen can be produced from 100.0g of potassium chlorate?...
Questions

History, 25.09.2019 22:50


Mathematics, 25.09.2019 22:50


Mathematics, 25.09.2019 22:50




History, 25.09.2019 22:50

Mathematics, 25.09.2019 22:50

Chemistry, 25.09.2019 22:50







Mathematics, 25.09.2019 22:50

Mathematics, 25.09.2019 22:50

Biology, 25.09.2019 22:50

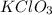 = 100.0 g
= 100.0 g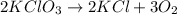
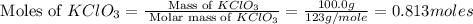
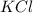 and
and 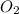 .
.

