
In this experiment, you will titrate a solution of hcl whose concentration is unknown with a solution of naoh whose concentration is known. based on what you just reviewed, express this goal in the form of an investigative question that you can answer by doing the experiment.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 2

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
In this experiment, you will titrate a solution of hcl whose concentration is unknown with a solutio...
Questions




History, 01.07.2019 12:30

Advanced Placement (AP), 01.07.2019 12:30

Mathematics, 01.07.2019 12:30



Mathematics, 01.07.2019 12:30


Mathematics, 01.07.2019 12:30

History, 01.07.2019 12:30


Advanced Placement (AP), 01.07.2019 12:30

Health, 01.07.2019 12:30


Mathematics, 01.07.2019 12:30

Mathematics, 01.07.2019 12:30


Mathematics, 01.07.2019 12:30

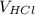 ×
× 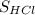 =
= 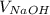 ×
× 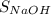 .
.

