Chemistry, 05.07.2019 11:00 nickname0097
Abaker uses sodium hydrogen carbonate (baking soda) as the leavening agent in a banana-nut quick bread. the baking soda decomposes according to two possible reactions. reaction 1: 2 nahco3(s) → na2co3(s) + h2o(l) + co2(g) reaction 2: nahco3(s) + h+(aq) → h2o(l) + co2(g) + na+(aq) calculate the volume (in ml) of co2 that forms at 170.°c and 0.900 atm per gram of nahco3 by each of the reaction processes.

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 07:30
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 23.06.2019 00:30
An unknown insoluble substance displaced the water shown. it's mass is indicated on the triple beam balance. mass = a. 694 b. 693.5 c. 693.0 d.693.8
Answers: 1
You know the right answer?
Abaker uses sodium hydrogen carbonate (baking soda) as the leavening agent in a banana-nut quick bre...
Questions




Biology, 06.11.2020 23:40

Mathematics, 06.11.2020 23:40

Mathematics, 06.11.2020 23:40


English, 06.11.2020 23:40

English, 06.11.2020 23:40


Mathematics, 06.11.2020 23:40


History, 06.11.2020 23:40



History, 06.11.2020 23:40

Geography, 06.11.2020 23:40

History, 06.11.2020 23:40

Social Studies, 06.11.2020 23:40

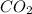 in Reaction 1 = 240.45 ml
in Reaction 1 = 240.45 ml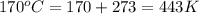 (
( )
)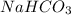 = 1 gram
= 1 gram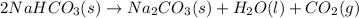


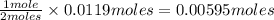 of
of