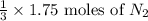Chemistry, 06.07.2019 01:00 biancabahena04
If you have 3.5 mol of n2, and 1.75 mol of h2, how many moles of n2 are there in excess? reaction: n2 + 3 h2 → 2 nh3

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 12:30
Wind is related tot he movement or warm cold air masses which kind of heat transfer does this represent
Answers: 2

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 07:30
In a reaction (at equilibrium) that makes more moles of gas than it consumes, what is the effect of increasing the pressure?
Answers: 1

You know the right answer?
If you have 3.5 mol of n2, and 1.75 mol of h2, how many moles of n2 are there in excess? reaction:...
Questions



Mathematics, 25.11.2019 20:31



Mathematics, 25.11.2019 20:31




Computers and Technology, 25.11.2019 20:31

Mathematics, 25.11.2019 20:31



History, 25.11.2019 20:31


Mathematics, 25.11.2019 20:31

Mathematics, 25.11.2019 20:31


Mathematics, 25.11.2019 20:31

Mathematics, 25.11.2019 20:31

 are present in excess.
are present in excess.
 = 1.75 moles
= 1.75 moles .
.