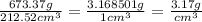Chemistry, 07.07.2019 01:30 mathlady12
Arectangular block of copper metals weigh 673.37g. the dimensions of the block are 8.4cm by 5.5cm by 4.6cm. from this data, what is the density of the copper?

Answers: 1
Another question on Chemistry


Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1

Chemistry, 23.06.2019 04:31
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1

Chemistry, 23.06.2019 08:10
Time remaining 58: 10 an atom that has 84 protons and 86 neutrons undergoes a reaction. at the end of the reaction, it has 82 protons and 84 neutrons. what happened to the atom? it accepted radiation in a chemical reaction it donated neutrons to another atom in a chemical reaction it emitted an alpha particle in a nuclear reaction. it accepted protons in a nuclear reaction. mark this and retum save and exit next submit
Answers: 3
You know the right answer?
Arectangular block of copper metals weigh 673.37g. the dimensions of the block are 8.4cm by 5.5cm by...
Questions

Business, 08.10.2019 11:00



Computers and Technology, 08.10.2019 11:00

Mathematics, 08.10.2019 11:00

Chemistry, 08.10.2019 11:00

English, 08.10.2019 11:00







Mathematics, 08.10.2019 11:00

Mathematics, 08.10.2019 11:00


Mathematics, 08.10.2019 11:00


Mathematics, 08.10.2019 11:00

Chemistry, 08.10.2019 11:00