
Chemistry, 07.07.2019 21:00 christi1175
The decomposition reaction of carbon disulfide to carbon monosulfide and sulfur is first order with k = 2.80 ✕ ✕ 10−7 sec-1 at 1000°c. cs2(g) → cs(g) + s(g) a. how much of a 4.83-gram sample of carbon disulfide would remain after 37.0 days? 1.97 1.97 grams carbon disulfide b. how much carbon monosulfide would be formed after 37.0 days? 1.14 1.65 grams carbon monosulfide useful information 1.013 bar = 760 torr = 1 atm = 760 mm hg

Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 23.06.2019 01:00
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1
You know the right answer?
The decomposition reaction of carbon disulfide to carbon monosulfide and sulfur is first order with...
Questions


Spanish, 07.05.2021 19:30

Business, 07.05.2021 19:30



Physics, 07.05.2021 19:30


Biology, 07.05.2021 19:30


Computers and Technology, 07.05.2021 19:30

Mathematics, 07.05.2021 19:30


Biology, 07.05.2021 19:30

English, 07.05.2021 19:30


Mathematics, 07.05.2021 19:30


Chemistry, 07.05.2021 19:30


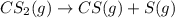
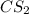 utilised in the reaction
utilised in the reaction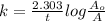
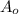 = Initial mass of reactant
= Initial mass of reactant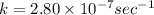
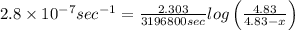
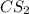 remained after 37 days = 4.83 - x
remained after 37 days = 4.83 - x

