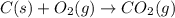The question: a new grill has a mass of 30.0 kg. you put 2.0 kg of charcoal in the grill. you burn all the charcoal and the grill has a mass of 30.0 kg. what is the mass in kg of the gases given off? (assume that the charcoal is pure carbon solid and that it burns completely in oxygen). the mass of carbon burnt = 2kg = 2000g moles of carbon burnt = 2000/12 => moles of co2 produced = 2000/12 => mass of co2 produced = 2000/12*44 = 2 333.33 g = 2.33 kg in the answer, where did the 44 come from?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2

Chemistry, 22.06.2019 23:30
What are the similarities between compounds and mixtures?
Answers: 3

Chemistry, 23.06.2019 00:10
Find the missing probability in the table below a.0.10 b.40 c.0.80 d. 0.20
Answers: 2
You know the right answer?
The question: a new grill has a mass of 30.0 kg. you put 2.0 kg of charcoal in the grill. you burn a...
Questions


English, 12.03.2021 17:20

Mathematics, 12.03.2021 17:20




Mathematics, 12.03.2021 17:20

Mathematics, 12.03.2021 17:20

History, 12.03.2021 17:20


Chemistry, 12.03.2021 17:20

Social Studies, 12.03.2021 17:20

Mathematics, 12.03.2021 17:20


Mathematics, 12.03.2021 17:20

Chemistry, 12.03.2021 17:20

Mathematics, 12.03.2021 17:20

Mathematics, 12.03.2021 17:20

Mathematics, 12.03.2021 17:20

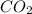 .
.