
Chemistry, 21.01.2020 13:31 VanBrocklin4706
If density is 22.57g/cm^3 what volume would be occupied by 4.80kg of osmium

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:00
19. at high pressures, how does the volume of a real gas compare with the volume of an ideal gas under the same conditions, and why? eman- it is much less because real gas partides are not moving. there is no difference because the gas laws are always obeyed. it is much less because at high pressures the temperature drops. it is much greater because real gas partides take up space.
Answers: 1

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1
You know the right answer?
If density is 22.57g/cm^3 what volume would be occupied by 4.80kg of osmium...
Questions


Mathematics, 30.11.2021 09:00


History, 30.11.2021 09:00


Biology, 30.11.2021 09:00

English, 30.11.2021 09:00

English, 30.11.2021 09:00





English, 30.11.2021 09:00


Mathematics, 30.11.2021 09:10

Mathematics, 30.11.2021 09:10

Advanced Placement (AP), 30.11.2021 09:10

Mathematics, 30.11.2021 09:10


Mathematics, 30.11.2021 09:10

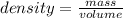
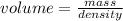
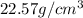
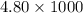 = 4800 g
= 4800 g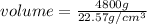
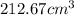 .
.

