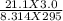Chemistry, 08.07.2019 19:30 taylor3865
Ahuman lung at maximum capacity has a volume of 3.0 liters. if the partial pressure of oxygen in the air is 21.1 kilopascals and the air temperature is 295 k, how many moles of oxygen are in the lung?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is important to study for nios grade 12 chemistry? i have only one month left.
Answers: 2

Chemistry, 22.06.2019 13:00
If two objects at different te,peraure are in contact with each other what happens to their temperature
Answers: 1

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have metallic bonds. which of the following is most likely a property of this substance? a. low conductivity b. low boiling point c. high malleability d. high solubility in water
Answers: 2
You know the right answer?
Ahuman lung at maximum capacity has a volume of 3.0 liters. if the partial pressure of oxygen in the...
Questions


Social Studies, 22.09.2019 17:50


Biology, 22.09.2019 17:50


Arts, 22.09.2019 17:50

History, 22.09.2019 17:50

Mathematics, 22.09.2019 17:50

History, 22.09.2019 17:50




Biology, 22.09.2019 17:50





Physics, 22.09.2019 17:50


Social Studies, 22.09.2019 17:50

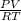 . On plugging the given values, we get,
. On plugging the given values, we get,