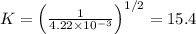Chemistry, 09.07.2019 01:00 sebasm4842
The equilibrium constant for the equilibrium, 3a+ 2b ↔ 2d + e, is 4.22 x 10-3 . what is the equilibrium constant for the equilibrium: d + (1/2)e ↔ (3/2)a + b? 2.1110-3237-2.1110-315.4

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3


Chemistry, 22.06.2019 10:40
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1
You know the right answer?
The equilibrium constant for the equilibrium, 3a+ 2b ↔ 2d + e, is 4.22 x 10-3 . what is the eq...
Questions


Mathematics, 22.10.2020 20:01



Mathematics, 22.10.2020 20:01




Mathematics, 22.10.2020 20:01


Mathematics, 22.10.2020 20:01


Mathematics, 22.10.2020 20:01


History, 22.10.2020 20:01


Mathematics, 22.10.2020 20:01